Goblet cells, mucus & mucosal defence

Our lab explores the mucosal surfaces that are the largest interface between the environment and our tissues. Their critical functions necessitate exposure of living cells to a vast collection of microbial lifeforms that includes many opportunistic and obligate pathogens. The epithelial cell layer strictly controls interactions between this environment and our internal systemic tissues. However, this single layer of cells can only endure in this setting due to the multi-layered defensive functions of the epithelial cellular lineages.
Our favourite epithelial lineage are the goblet cells that specialise in the synthesis and secretion of gel-forming mucins and other mucus components that restrict microbial access to the epithelium. Historically considered a single, unifunctional cell type, our research shows that they comprise multiple, functionally diverse sub-populations that contribute to different aspects of mucosal defence, thereby regulating our interactions with the mucosal environment.
In collaboration with the wider Mucin Biology Groups cluster at the University of Gothenburg and both national and international partners, our laboratory seeks to develop new tools allowing the identification and characterisation of goblet cell-mediated defence systems. Our goal is to understand their functions throughout the human lifespan, from pregnancy and birth to early life and ageing and to define their roles in the pathoaetiology of mucosal-related diseases including pre-term birth, infection, colorectal cancer and inflammatory bowel disease, with the ultimate purpose of developing innovative prophylactic and therapeutic strategies.
Current team

George Birchenough - Associate Professor/PI
George is a research group leader and tenured faculty at the department of Medical Biochemistry & Cell Biology at Sahlgrenska Academy, University of Gothenburg. Originally trained as a medical microbiologist working with neonatal infection at University College London, he went on to complete a postdoc investigating mucus and goblet cell biology in Gunnar Hansson’s Mucin Biology Group at the University of Gothenburg. In 2020 he was appointed as a Research Fellow at the Wallenberg Centre for Molecular & Translational Medicine. George’s CV can be found here

Mahadevan V. Subramani - Postdoctoral Researcher
Dev joined the lab as our first PhD student in 2021 after completing his MSc thesis work at the Karolinska Institute. His project examined the interactions of bacterial pathogens with intestinal goblet cell subtypes, and was successfully defended in December 2025.
He is currently working as a postdoctoral researcher to complete his doctoral research and develop his own independent research program.

Karl Hansson - Senior Researcher
Karl joined the lab as a postdoc in 2021 after working as a consultant for AstraZeneca and completing a PhD in neuroscience at the University of Gothenburg. His project aims to determine the role of goblet cell-intrinsic defence failure in the development of colorectal cancer.
He continues this role as a Senior Researcher in the group, coordinating the pre-clinical and translational CRC research in the lab.

Alex Bennett - Postdoctoral Researcher
Alex joined the lab as a joint postdoc with Daniel Bojar’s lab in 2023 after completing his PhD in Immunology at the University of Manchester. His project applies custom bioinformatics and multi-omic analysis to examine how diurnal/circadian alterations in mucus properties, especially mucin O-glycosylation, regulate host-microbiome/pathogen interactions in the gut.

Joana K Volk - PhD student
Joana first joined the lab as an Erasmus student in 2017, where she worked on a project defining the role of inflammasomes in intestinal mucus barrier development.
After completing her MSc in Molecular Medicine she went on to complete an MD at the University of Freiburg, after which she returned to Gothenburg to undertake specialist training in obstetrics and gynecology at Sahlgrenska University Hospital.
Joana rejoined the lab as a part-time PhD student in 2025, where she works on a project to characterise cervical mucus plug molecular properties in pregnancy and to identify associations with intra-uterine infection and preterm labour.

Katharine Williams - PhD student
After completing her MSc in Molecular Medicine at the University of East Anglia, Kat first joined the lab as a Research Assistant in 2024 where she worked on a project to define the role of short chain fatty acid sensing by intestinal goblet cells in regulating host-microbe interactions.
She was awarded a PhD position in the lab in 2025, where she continues to work on mucus-microbe interactions in the gut, specifically how early life development of goblet cell-intrinsic protection is important in modulating the gut-brain axis and life-long susceptibility to disease.

Setu Dambhare - Research Assistant/PhD candidate
After completing an MSc in Molecular Biology, Molecular Genetics and Biotechnology jointly at Lund University and AstraZeneca, Setu joined the lab as a Research Assistant and prospective PhD student in 2025.
Setu’s project is to define vagino-cervical goblet cell and mucus properties and functions during estrus cycling and pregnancy in pre-clinical mouse models, with the goal to employ such models to study host-microbe interactions in healthy and preterm pregnancy.

Lab alumni:
Åsa Johansson - Doctoral candidate (co-supervised)
Åsa joined the lab as an associated PhD student working under the supervision of Dr. Malin Johansson. She worked part time as a Bitr. Verksamhetschef in Clinical Microbiology in Växjö, and part time in studying how goblet cell-intrinsic defences develop during the post-natal period.
After her research, Åsa has pursued a career in industry and currently works as CEO for Offspring Biosciences

Juan Sienes - Erasmus student
Juan joined the lab in 2021 as an Erasmus project student, working to establish new methods in accurately quantifying goblet cell dynamics during infection and inflammation.
After his time in the lab, Juan was offered a PhD position at the Pasteur Institute in Paris.

Akshi Singla - Postdoctoral researcher
Akshi joined the lab as a postdoc in 2022 after working as a postdoc in the microbiology and immunology department at University of Gothenburg and completing a PhD in chemical engineering at Texas A&M University. Her project aimed at identifying specific microbiota-host signaling pathways that regulate goblet cell dependent host microbiome interactions.
After completing her postdoctoral work - Akshi received an Assistant Professor position at the Delhi Institute of Technology where she is now establishing her own laboratory.
Our research
Sharba S, Sundh H, Santos L, Sundell K, Birchenough G, Dalum AS, Loibman S, Benktander J, Lindén SK. A. salmonicida infection inhibits rainbow trout gill mucin production. Fish Shellfish Immunol. 2025 Dec 22;:111089. doi: 10.1016/j.fsi.2025.111089. [Epub ahead of print] PubMed PMID: 41443520.
Jakob MO, Sterczyk N, Boulekou S, Forster PM, Barleben L, Alzain N, Jarick KJ, Pirzgalska RM, Raposo B, Hansson K, Nyström EEL, Gondrand A, González-Acera M, Leclère PS, Lapson MS, Poggenseier S, Deshpande D, Velleman L, Breiderhoff T, Brunkhorst MF, Schüle AM, Guerra GM, Durek P, Mashreghi MF, Kühl AA, Chu C, Schneider C, Weidinger C, Siegmund B, Nordmann TM, Voehringer D, Patankar JV, Becker C, Birchenough GMH, Veiga-Fernandes H, Ronchi F, Kolesnichenko M, Diefenbach A, Klose CSN. Enteric nervous system-derived VIP restrains differentiation of LGR5(+) stem cells toward the secretory lineage impeding type 2 immune programs. Nat Immunol. 2025 Dec;26(12):2227-2243. doi: 10.1038/s41590-025-02325-1. Epub 2025 Nov 24. PubMed PMID: 41286457.
Yilmaz B, Baertschi I, Meier KHU, Le Gac C, Jordi SBU, Black C, Li J, Lindholm AK, König B, Sauer U, Stelling J, Macpherson AJ. A global survey of taxa-metabolic associations across mouse microbiome communities. Cell Host Microbe. 2025 Nov 12;33(11):1960-1976.e10. doi: 10.1016/j.chom.2025.10.010. Epub 2025 Nov 3. PubMed PMID: 41187758.
Johansson Å, Venkita Subramani M, Yilmaz B, Nyström EEL, Layunta E, Arike L, Sommer F, Rosenstiel P, Vereecke L, Mannerås-Holm L, Wullaert A, Pelaseyed T, Johansson MEV, Birchenough GMH. Neonatal microbiota colonization primes maturation of goblet cell-mediated protection in the pre-weaning colon. J Exp Med. 2025 Aug 4;222(8). doi: 10.1084/jem.20241591. Epub 2025 May 5. PubMed PMID: 40323318; PubMed Central PMCID: PMC12051479.
Winsor NJ, Bayer G, Singh O, Chan JK, Li LY, Lieng BY, Foerster E, Popovic A, Tsankov BK, Maughan H, Lemire P, Tam E, Streutker C, Chen L, Heaver SL, Ley RE, Parkinson J, Montenegro-Burke JR, Birchenough GMH, Philpott DJ, Girardin SE. Cross-kingdom-mediated detection of intestinal protozoa through NLRP6. Cell Host Microbe. 2025 Mar 12;33(3):388-407.e9. doi: 10.1016/j.chom.2025.02.008. Epub 2025 Mar 4. PubMed PMID: 40043701.
Azimzadeh PN, Birchenough GM, Gualbuerto NC, Pinkner JS, Tamadonfar KO, Beatty W, Hannan TJ, Dodson KW, Ibarra EC, Kim S, Schreiber HL 4th, Janetka JW, Kau AL, Earl AM, Miller MJ, Hansson GC, Hultgren SJ. Mechanisms of uropathogenic E. coli mucosal association in the gastrointestinal tract. Sci Adv. 2025 Jan 31;11(5):eadp7066. doi: 10.1126/sciadv.adp7066. Epub 2025 Jan 31. PubMed PMID: 39888987; PubMed Central PMCID: PMC11784811.
(Preprint) Johansson Å, Subramani MV, Yilmaz B, Nyström E, Layunta E, Arike L, Sommer F, Rosenstiel P, Vereecke L, Holm LM, Wullaert A, Pelaseyed T, Johansson MEV, Birchenough GMH. Neonatal microbiota colonization drives maturation of primary and secondary goblet cell mediated protection in the pre-weaning colon. bioRxiv. 2024 Jul 5;. doi: 10.1101/2024.07.03.601781. PubMed PMID: 39005291; PubMed Central PMCID: PMC11245021.
van Gorp C, de Lange IH, Hütten MC, López-Iglesias C, Massy KRI, Kessels L, Knoops K, Cuijpers I, Sthijns MMJPE, Troost FJ, van Gemert WG, Spiller OB, Birchenough GMH, Zimmermann LJI, Wolfs TGAM. Antenatal Ureaplasma Infection Causes Colonic Mucus Barrier Defects: Implications for Intestinal Pathologies. Int J Mol Sci. 2024 Apr 3;25(7). doi: 10.3390/ijms25074000. PubMed PMID: 38612809; PubMed Central PMCID: PMC11011967.
Arnesen H, Markussen T, Birchenough G, Birkeland S, Nyström EEL, Hansson GC, Carlsen H, Boysen P. Microbial experience through housing in a farmyard-type environment alters intestinal barrier properties in mouse colons. Sci Rep. 2023 Aug 22;13(1):13701. doi: 10.1038/s41598-023-40640-5. PubMed PMID: 37607995; PubMed Central PMCID: PMC10444815.
Devarakonda S, Thorsell A, Hedenström P, Rezapour A, Heden L, Banerjee S, Johansson MEV, Birchenough G, Toft Morén A, Gustavsson K, Skokic V, Pettersson VL, Sjöberg F, Kalm M, Al Masri M, Ekh M, Fagman H, Wolving M, Perkins R, Morales RA, Castillo F, Villablanca EJ, Yrlid U, Bergmark K, Steineck G, Bull C. Low-grade intestinal inflammation two decades after pelvic radiotherapy. EBioMedicine. 2023 Aug;94:104691. doi: 10.1016/j.ebiom.2023.104691. Epub 2023 Jul 20. PubMed PMID: 37480626; PubMed Central PMCID: PMC10393618.
Song R, McAlpine W, Fond AM, Nair-Gill E, Choi JH, Nyström EEL, Arike L, Field S, Li X, SoRelle JA, Moresco JJ, Moresco EMY, Yates JR 3rd, Azadi P, Ni J, Birchenough GMH, Beutler B, Turer EE. Trans-Golgi protein TVP23B regulates host-microbe interactions via Paneth cell homeostasis and Goblet cell glycosylation. Nat Commun. 2023 Jun 20;14(1):3652. doi: 10.1038/s41467-023-39398-1. PubMed PMID: 37339972; PubMed Central PMCID: PMC10282085.
Birchenough GMH*, Schroeder BO, Sharba S, Arike L, Recktenwald CV, Puértolas-Balint F, Subramani MV, Hansson KT, Yilmaz B, Lindén SK, Bäckhed F, Hansson GC. Muc2-dependent microbial colonization of the jejunal mucus layer is diet sensitive and confers local resistance to enteric pathogen infection. Cell Rep. 2023 Feb 28;42(2):112084. doi: 10.1016/j.celrep.2023.112084. Epub 2023 Feb 6. PubMed PMID: 36753416; PubMed Central PMCID: PMC10404306. (*corresponding author)
van Gorp C, de Lange IH, Hütten MC, López-Iglesias C, Massy KR, Kessels L, Kramer B, van de Wetering W, Spiller B, Birchenough GM, van Gemert WG, Zimmermann LJ, Wolfs TG. Antenatal Ureaplasma infection induces ovine small intestinal goblet cell defects: a strong link with NEC pathology. Tissue Barriers. 2023 Oct 2;11(4):2158016. doi: 10.1080/21688370.2022.2158016. Epub 2022 Dec 28. PubMed PMID: 36576242; PubMed Central PMCID: PMC10606782.
Sharba S, Sundh H, Sundell K, Benktander J, Santos L, Birchenough G, Lindén SK. Rainbow trout gastrointestinal mucus, mucin production, mucin glycosylation and response to lipopolysaccharide. Fish Shellfish Immunol. 2022 Mar;122:181-190. doi: 10.1016/j.fsi.2022.01.031. Epub 2022 Jan 22. PubMed PMID: 35077869.
Sharpen JDA, Dolan B, Nyström EEL, Birchenough GMH, Arike L, Martinez-Abad B, Johansson MEV, Hansson GC, Recktenwald CV. Transglutaminase 3 crosslinks the secreted gel-forming mucus component Mucin-2 and stabilizes the colonic mucus layer. Nat Commun. 2022 Jan 11;13(1):45. doi: 10.1038/s41467-021-27743-1. PubMed PMID: 35017479; PubMed Central PMCID: PMC8752817.
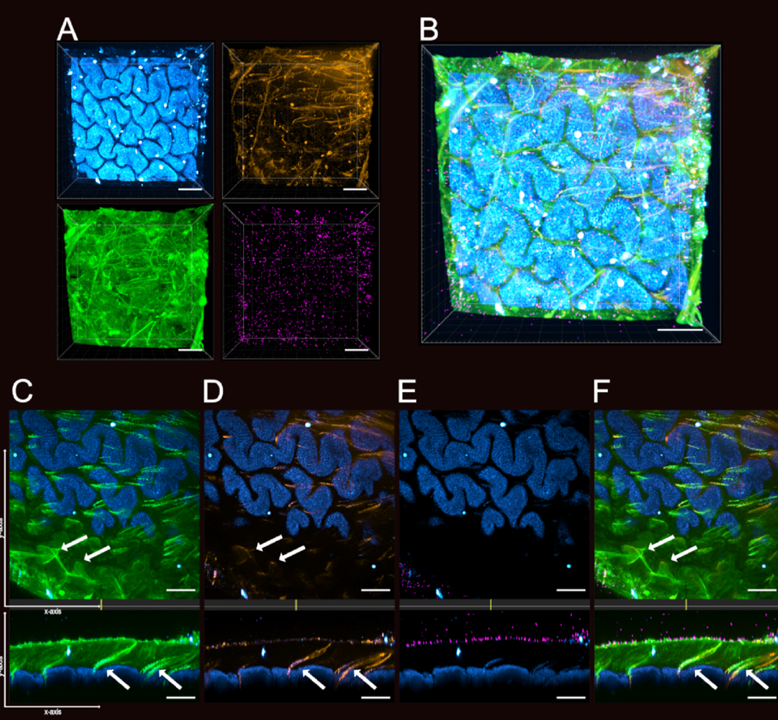
Nyström EEL, Martinez-Abad B, Arike L, Birchenough GMH, Nonnecke EB, Castillo PA, Svensson F, Bevins CL, Hansson GC, Johansson MEV. An intercrypt subpopulation of goblet cells is essential for colonic mucus barrier function. Science. 2021 Apr 16;372(6539). doi: 10.1126/science.abb1590. PubMed PMID: 33859001.
Benktander J, Padra JT, Maynard B, Birchenough G, Botwright NA, McCulloch R, Wynne JW, Sharba S, Sundell K, Sundh H, Lindén SK. Gill Mucus and Gill Mucin O-glycosylation in Healthy and Amebic Gill Disease-Affected Atlantic Salmon. Microorganisms. 2020 Nov 26;8(12). doi: 10.3390/microorganisms8121871. PubMed PMID: 33256221; PubMed Central PMCID: PMC7768351.
Birchenough GMH, Johansson MEV. Forming a mucus barrier along the colon. Science. 2020 Oct 23;370(6515):402-403. doi: 10.1126/science.abe7194. PubMed PMID: 33093095.
Schroeder BO, Birchenough GMH, Pradhan M, Nyström EEL, Henricsson M, Hansson GC, Bäckhed F. Obesity-associated microbiota contributes to mucus layer defects in genetically obese mice. J Biol Chem. 2020 Nov 13;295(46):15712-15726. doi: 10.1074/jbc.RA120.015771. Epub 2020 Sep 8. PubMed PMID: 32900852; PubMed Central PMCID: PMC7667970.
van der Post S, Jabbar KS, Birchenough G, Arike L, Akhtar N, Sjovall H, Johansson MEV, Hansson GC. Structural weakening of the colonic mucus barrier is an early event in ulcerative colitis pathogenesis. Gut. 2019 Dec;68(12):2142-2151. doi: 10.1136/gutjnl-2018-317571. Epub 2019 Mar 26. PubMed PMID: 30914450; PubMed Central PMCID: PMC6872445.
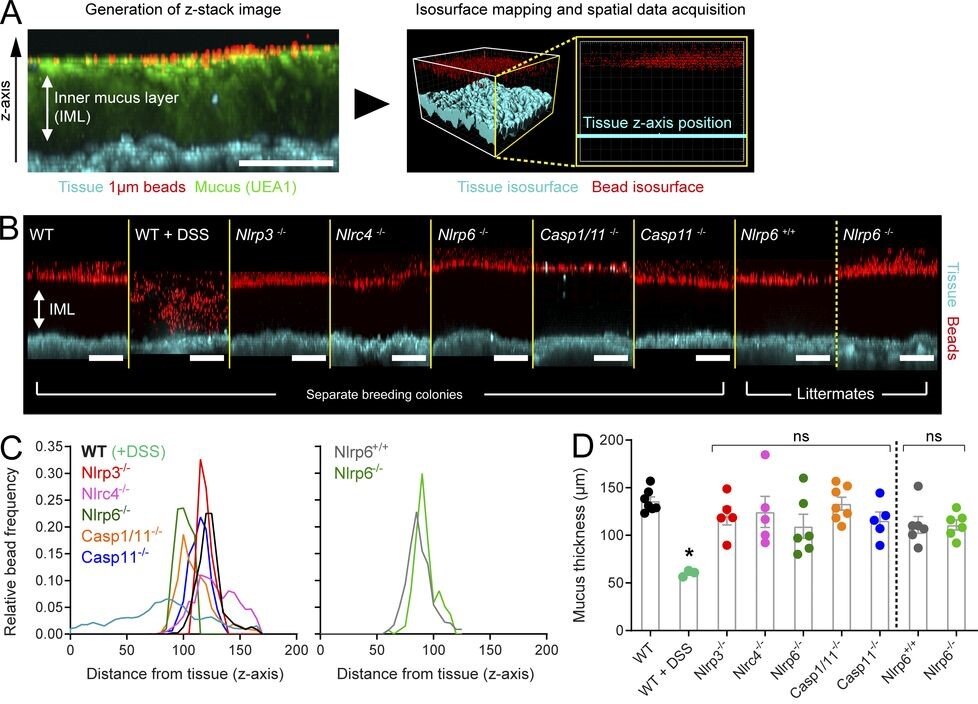
Volk JK, Nyström EEL, van der Post S, Abad BM, Schroeder BO, Johansson Å, Svensson F, Jäverfelt S, Johansson MEV, Hansson GC, Birchenough GMH. The Nlrp6 inflammasome is not required for baseline colonic inner mucus layer formation or function. J Exp Med. 2019 Nov 4;216(11):2602-2618. doi: 10.1084/jem.20190679. Epub 2019 Aug 16. PubMed PMID: 31420376; PubMed Central PMCID: PMC6829596.
McCarthy AJ, Birchenough GMH, Taylor PW. Loss of Trefoil Factor 2 Sensitizes Rat Pups to Systemic Infection with the Neonatal Pathogen Escherichia coli K1. Infect Immun. 2019 Mar;87(5). doi: 10.1128/IAI.00878-18. Print 2019 Mar. PubMed PMID: 30833331; PubMed Central PMCID: PMC6479038.
Birchenough G, Schroeder BO, Bäckhed F, Hansson GC. Dietary destabilisation of the balance between the microbiota and the colonic mucus barrier. Gut Microbes. 2019;10(2):246-250. doi: 10.1080/19490976.2018.1513765. Epub 2018 Sep 25. PubMed PMID: 30252606; PubMed Central PMCID: PMC6546334.
Nyström EEL, Birchenough GMH, van der Post S, Arike L, Gruber AD, Hansson GC, Johansson MEV. Calcium-activated Chloride Channel Regulator 1 (CLCA1) Controls Mucus Expansion in Colon by Proteolytic Activity. EBioMedicine. 2018 Jul;33:134-143. doi: 10.1016/j.ebiom.2018.05.031. Epub 2018 Jun 7. PubMed PMID: 29885864; PubMed Central PMCID: PMC6085540.
Schroeder BO, Birchenough GMH, Ståhlman M, Arike L, Johansson MEV, Hansson GC, Bäckhed F. Bifidobacteria or Fiber Protects against Diet-Induced Microbiota-Mediated Colonic Mucus Deterioration. Cell Host Microbe. 2018 Jan 10;23(1):27-40.e7. doi: 10.1016/j.chom.2017.11.004. Epub 2017 Dec 21. PubMed PMID: 29276171; PubMed Central PMCID: PMC5764785.
Birchenough G, Hansson GC. Bacteria Tell Us How to Protect Our Intestine. Cell Host Microbe. 2017 Jul 12;22(1):3-4. doi: 10.1016/j.chom.2017.06.011. PubMed PMID: 28704650.
Bosmans JW, Jongen AC, Birchenough GM, Nyström EE, Gijbels MJ, Derikx JP, Bouvy ND, Hansson GC. Functional mucous layer and healing of proximal colonic anastomoses in an experimental model. Br J Surg. 2017 Apr;104(5):619-630. doi: 10.1002/bjs.10456. Epub 2017 Feb 13. PubMed PMID: 28195642.
Birchenough GM*, Dalgakiran F*, Witcomb LA, Johansson ME, McCarthy AJ, Hansson GC, Taylor PW. Postnatal development of the small intestinal mucosa drives age-dependent, regio-selective susceptibility to Escherichia coli K1 infection. Sci Rep. 2017 Mar 6;7(1):83. doi: 10.1038/s41598-017-00123-w. PubMed PMID: 28250440; PubMed Central PMCID: PMC5427930. (* shared first author)
Bergström JH*, Birchenough GM*, Katona G, Schroeder BO, Schütte A, Ermund A, Johansson ME, Hansson GC. Gram-positive bacteria are held at a distance in the colon mucus by the lectin-like protein ZG16. Proc Natl Acad Sci U S A. 2016 Nov 29;113(48):13833-13838. doi: 10.1073/pnas.1611400113. Epub 2016 Nov 14. PubMed PMID: 27849619; PubMed Central PMCID: PMC5137749. (* shared first author)
Birchenough GM, Nyström EE, Johansson ME, Hansson GC. A sentinel goblet cell guards the colonic crypt by triggering Nlrp6-dependent Muc2 secretion. Science. 2016 Jun 24;352(6293):1535-42. doi: 10.1126/science.aaf7419. PubMed PMID: 27339979; PubMed Central PMCID: PMC5148821.
Erickson NA, Nyström EE, Mundhenk L, Arike L, Glauben R, Heimesaat MM, Fischer A, Bereswill S, Birchenough GM, Gruber AD, Johansson ME. The Goblet Cell Protein Clca1 (Alias mClca3 or Gob-5) Is Not Required for Intestinal Mucus Synthesis, Structure and Barrier Function in Naive or DSS-Challenged Mice. PLoS One. 2015;10(7):e0131991. doi: 10.1371/journal.pone.0131991. eCollection 2015. PubMed PMID: 26162072; PubMed Central PMCID: PMC4498832.
Birchenough GM, Johansson ME, Gustafsson JK, Bergström JH, Hansson GC. New developments in goblet cell mucus secretion and function. Mucosal Immunol. 2015 Jul;8(4):712-9. doi: 10.1038/mi.2015.32. Epub 2015 Apr 15. Review. PubMed PMID: 25872481; PubMed Central PMCID: PMC4631840.
Dalgakiran F, Witcomb LA, McCarthy AJ, Birchenough GM, Taylor PW. Non-invasive model of neuropathogenic Escherichia coli infection in the neonatal rat. J Vis Exp. 2014 Oct 29;(92):e52018. doi: 10.3791/52018. PubMed PMID: 25408299; PubMed Central PMCID: PMC4353393.
Pelaseyed T, Bergström JH, Gustafsson JK, Ermund A, Birchenough GM, Schütte A, van der Post S, Svensson F, Rodríguez-Piñeiro AM, Nyström EE, Wising C, Johansson ME, Hansson GC. The mucus and mucins of the goblet cells and enterocytes provide the first defense line of the gastrointestinal tract and interact with the immune system. Immunol Rev. 2014 Jul;260(1):8-20. doi: 10.1111/imr.12182. Review. PubMed PMID: 24942678; PubMed Central PMCID: PMC4281373.
Birchenough GM, Johansson ME, Stabler RA, Dalgakiran F, Hansson GC, Wren BW, Luzio JP, Taylor PW. Altered innate defenses in the neonatal gastrointestinal tract in response to colonization by neuropathogenic Escherichia coli. Infect Immun. 2013 Sep;81(9):3264-75. doi: 10.1128/IAI.00268-13. Epub 2013 Jun 24. PubMed PMID: 23798529; PubMed Central PMCID: PMC3754193.
Dawson LF, Valiente E, Donahue EH, Birchenough G, Wren BW. Hypervirulent Clostridium difficile PCR-ribotypes exhibit resistance to widely used disinfectants. PLoS One. 2011;6(10):e25754. doi: 10.1371/journal.pone.0025754. Epub 2011 Oct 25. PubMed PMID: 22039420; PubMed Central PMCID: PMC3201945.
Zelmer A, Martin MJ, Gundogdu O, Birchenough G, Lever R, Wren BW, Luzio JP, Taylor PW. Administration of capsule-selective endosialidase E minimizes upregulation of organ gene expression induced by experimental systemic infection with Escherichia coli K1. Microbiology. 2010 Jul;156(Pt 7):2205-2215. doi: 10.1099/mic.0.036145-0. Epub 2010 Apr 15. PubMed PMID: 20395269; PubMed Central PMCID: PMC2923034.
Selected Publications
2025
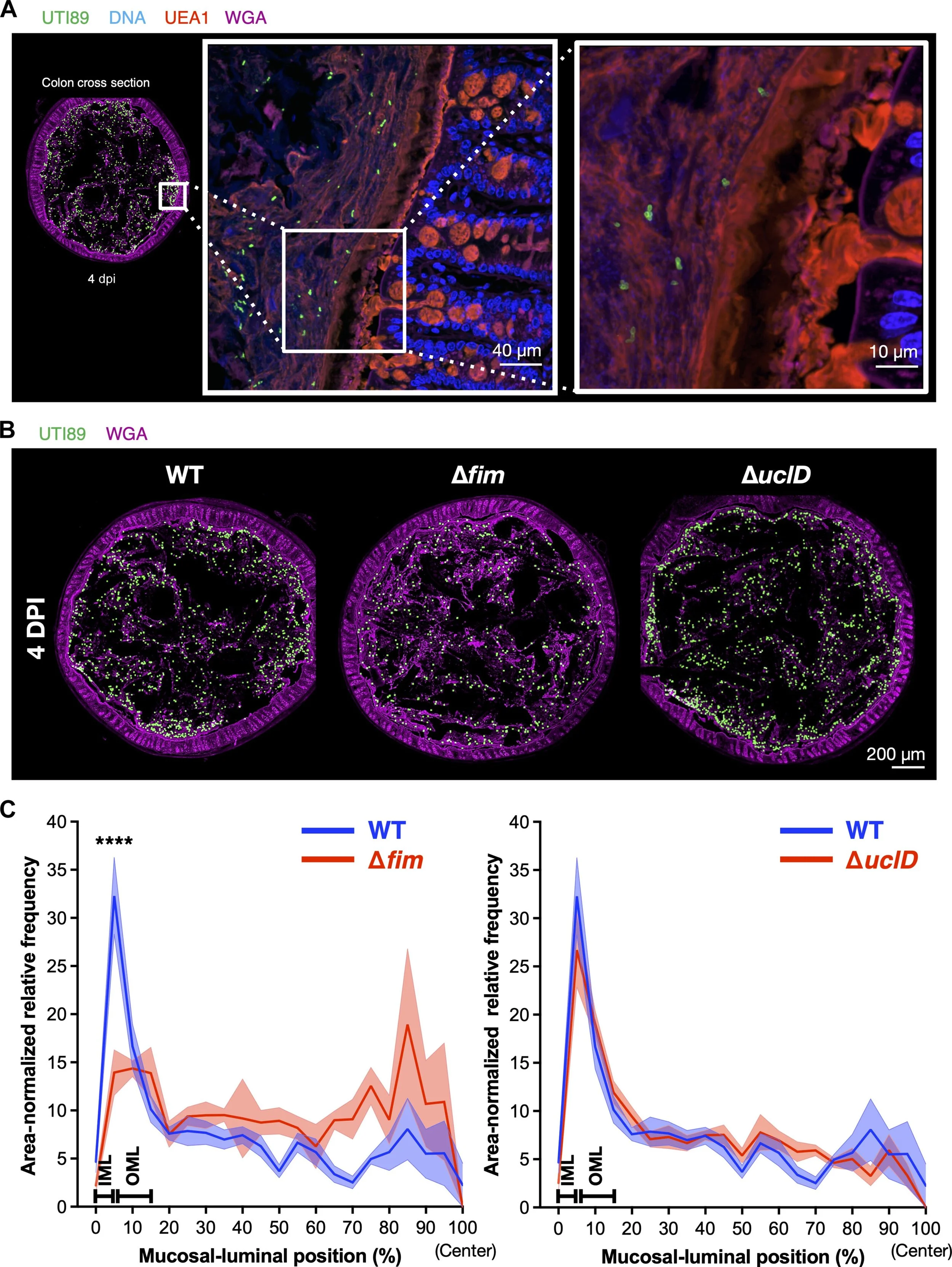
Mechanisms of uropathogenic E. coli mucosal association in the gastrointestinal tract
Collaboration with Scott Hultgren’s lab at Washington University published in Science Advances differentiating the roles of UPEC adhesin proteins in determining the spatiotemporal dynamics of intestinal colonisation.
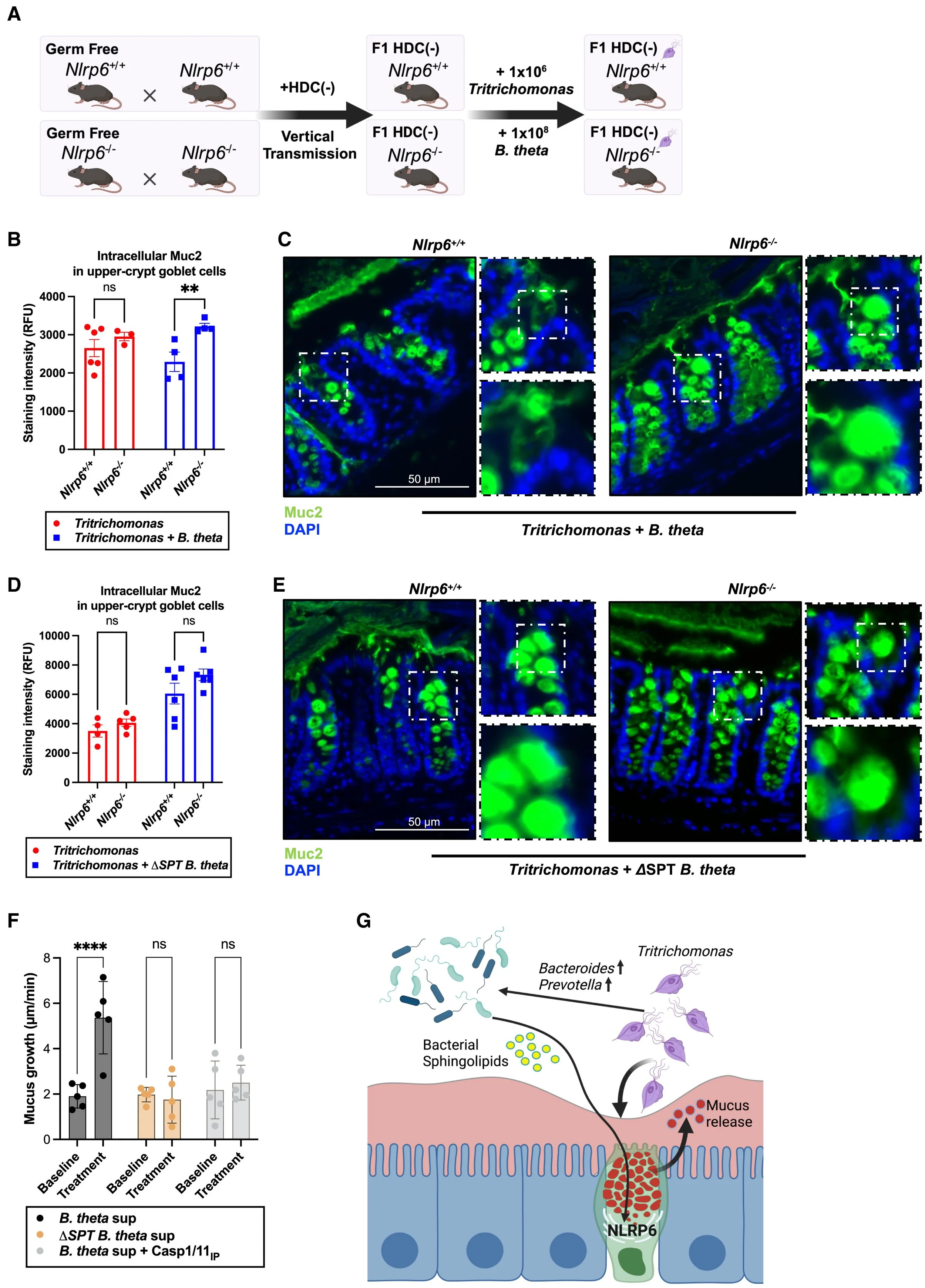
Cross-kingdom-mediated detection of intestinal protozoa through NLRP6
Collaboration with the labs of Stephen Girardin and Dana Philpott at the University of Toronto published in Cell Host & Microbe that identifies a novel interaction between intestinal protozoa, bacteria and sentinel goblet cells.
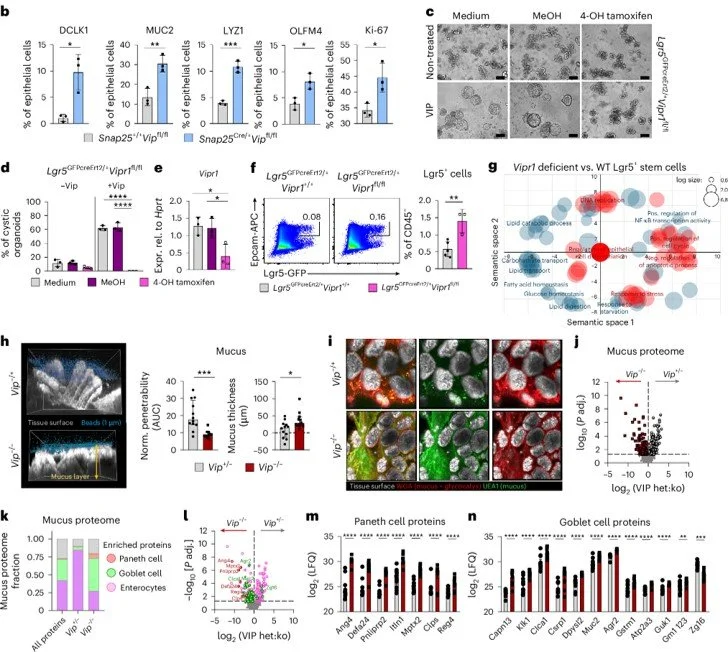
Enteric nervous system-derived VIP restrains differentiation of LGR5+ stem cells toward the secretory lineage impeding type 2 immune programs
Collaboration with Christoph Klose’s lab at Charité - Universitätsmedizin Berlin published in Nature Immunology dissects an enteric nervous system - epithelial signalling pathway which regulates the differentiation of secretory cells and induction of type 2 immunity.
2024
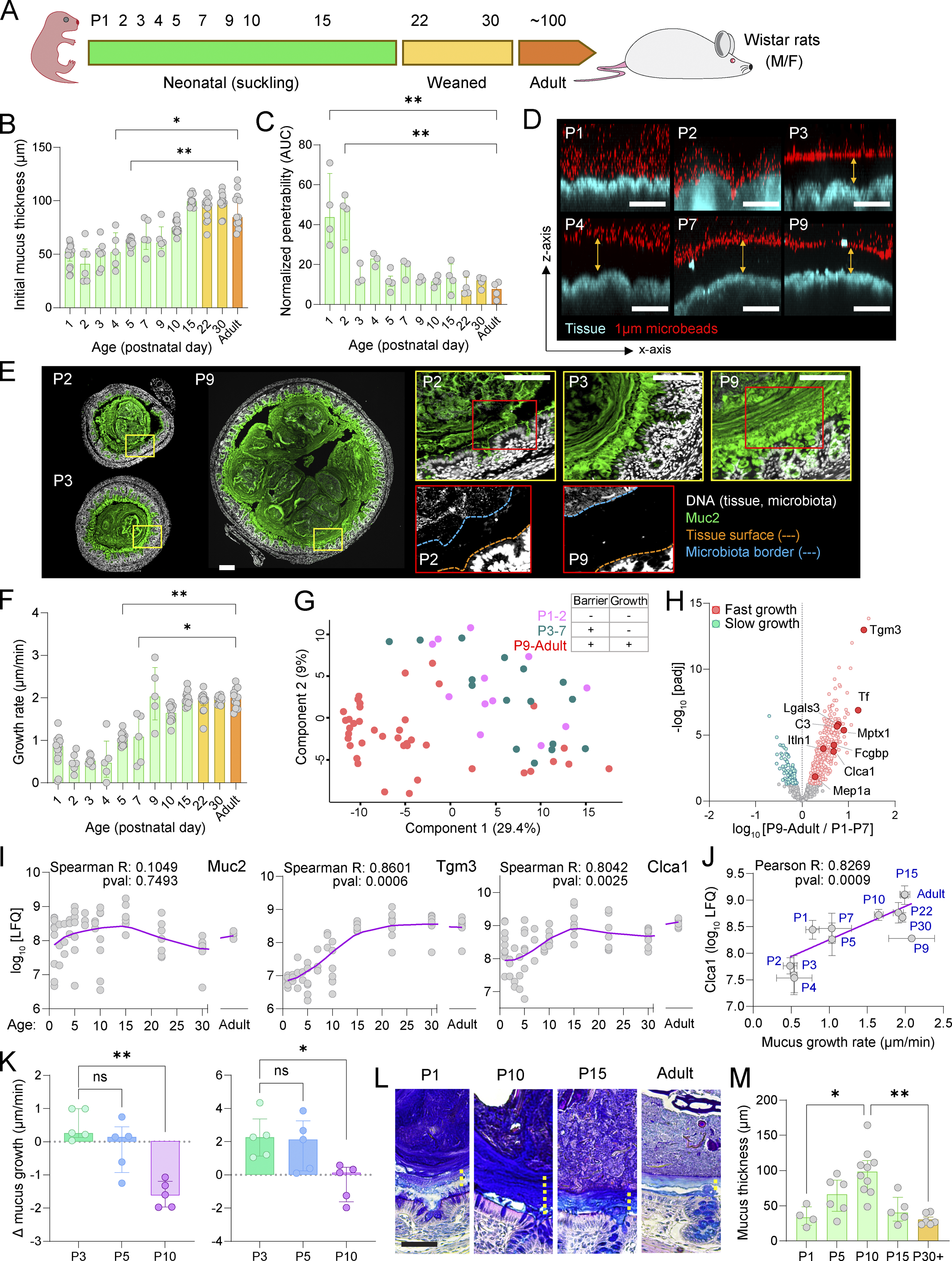
Neonatal microbiota colonization primes maturation of goblet cell-mediated protection in the pre-weaning colon
Preprint from our lab (subsequently published in Journal of Experimental Medicine) providing the first in depth characterisation of postnatal colonic mucus barrier development and priming of the sentinel goblet cell dependent secretory response by the microbiome.
2023
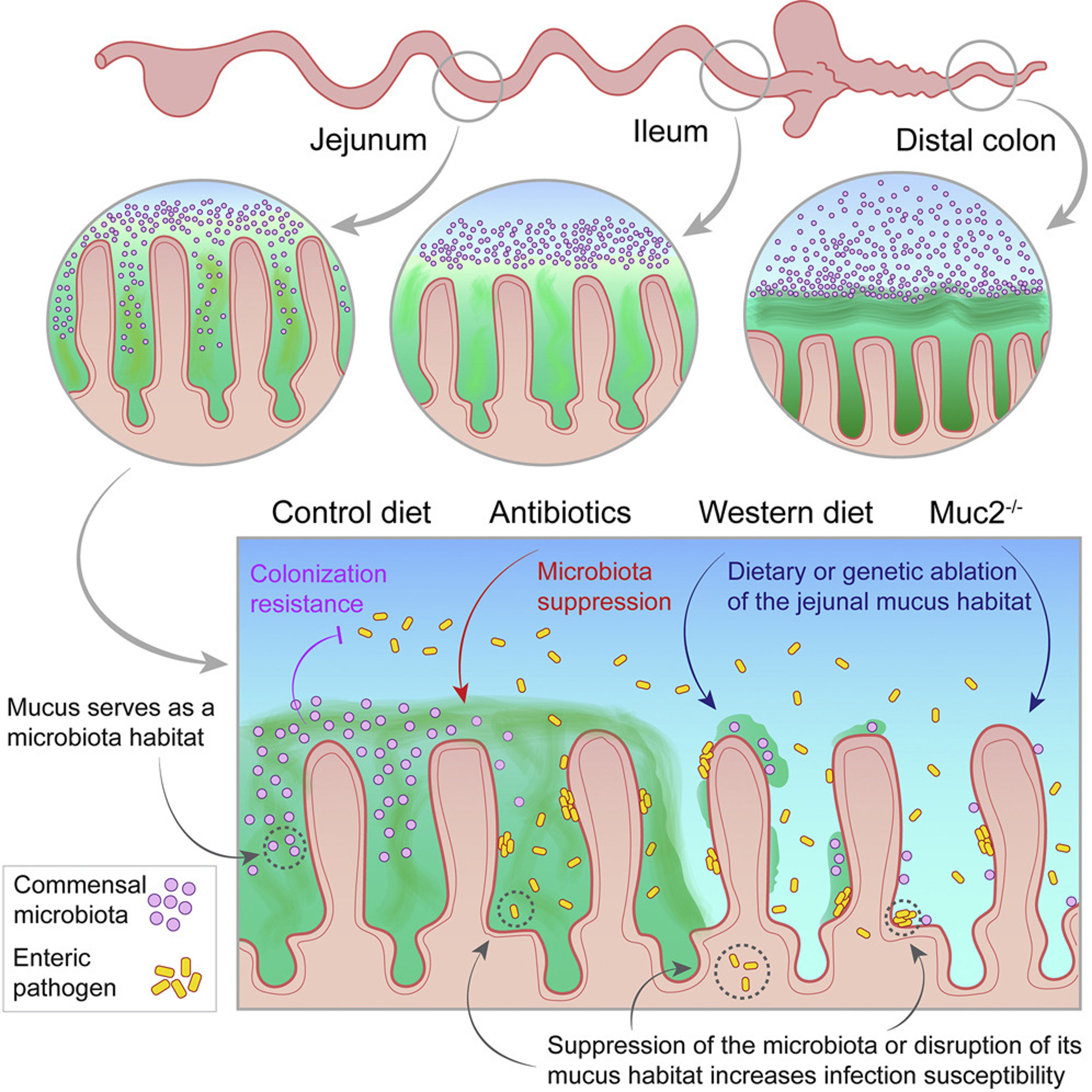
Muc2-dependent microbial colonization of the jejunal mucus layer is diet sensitive and confers local resistance to enteric pathogen infection
Study from our lab published in Cell Reports identifying a novel role for jejunal mucus in supporting colonisation resistance in the small intestine. Our work provides evidence of mucus acting as a habitat for protective bacteria, rather than acting solely as a barrier mechanism.
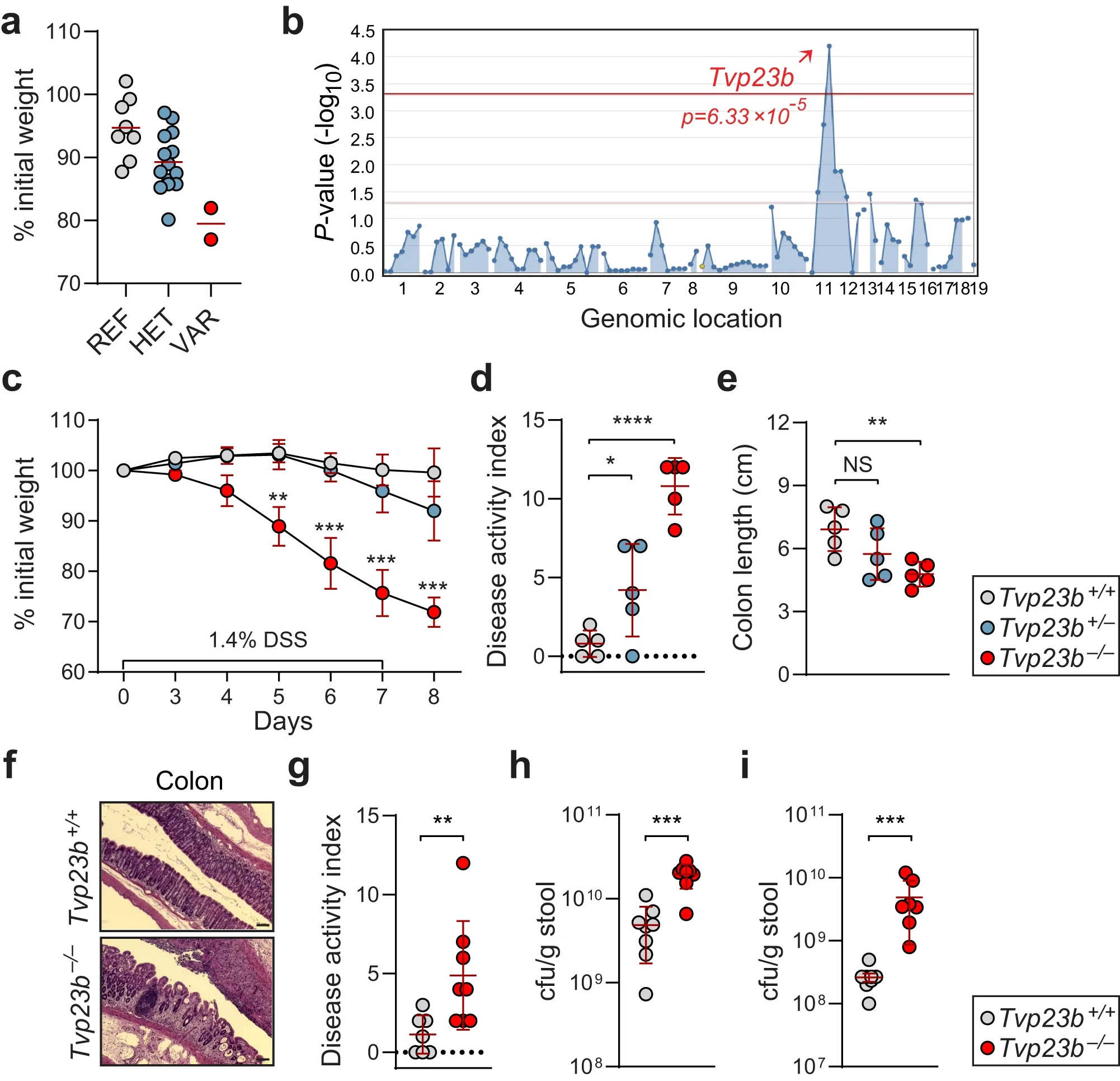
Trans-Golgi protein TVP23B regulates host-microbe interactions via Paneth cell homeostasis and Goblet cell glycosylation
Collaboration with Emre Turer’s lab at UT Southwestern published in Nature Communications investigating the pivotal function of Tvp23b in intestinal secretory cells.
2022
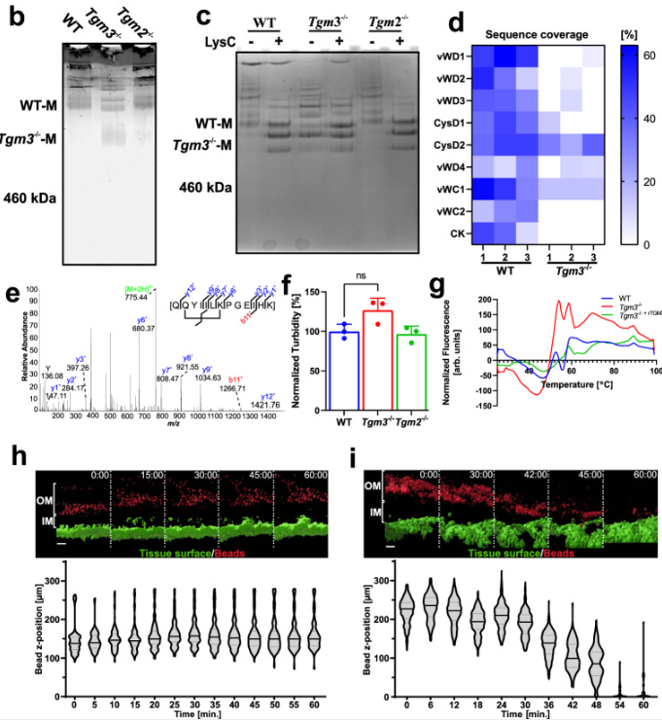
Transglutaminase 3 crosslinks the secreted gel-forming mucus component Mucin-2 and stabilizes the colonic mucus layer
Collaboration with fellow Mucin Biology Groups researcher Christian Recktenwald published in Nature Communications establishing a crucial role for Tgm3 catalysed Muc2 crosslinking in preventing microbial proteolysis of intestinal mucus.
Rainbow trout gastrointestinal mucus, mucin production, mucin glycosylation and response to lipopolysaccharide.
Collaboration with Sara Linden’s lab published in Fish & Shellfish Immunology examining in vivo mucus barrier properties of the trout gastrointestinal tract.

2021
NOX1-dependent redox signaling potentiates colonic stem cell proliferation to adapt to the intestinal microbiota by linking EGFR and TLR activation
Collaboration with Sjoerd van der Post published in Cell Reports demonstrating the key role of Nox1 in linking microbial components to epithelial stem cell proliferation.

An intercrypt subpopulation of goblet cells is essential for colonic mucus barrier function
Collaboration with Malin Johansson’s lab published in Science identifying the critical role of intercrypt goblet cells (icGCs) in maintaining colonic mucus barrier structure.
2020

Obesity-associated microbiota contributes to mucus layer defects in genetically obese mice
Collaboration with Björn Schröder’s lab at Umeå University published in JBC, identifying colonic mucus barrier defects in genetically obese mice with causal links to microbiome composition.

Perspective in Science on Kirk Bergstrom and Lijun Xia’s paper describing how mucus produced in the proximal colon contributes to distal colonic barrier function.
2019
Structural weakening of the colonic mucus barrier is an early event in ulcerative colitis pathogenesis
Collaboration with Gunnar Hansson’s lab published in Gut reveals goblet cell-intrinsic defence system dysfunction in UC patients
Loss of Trefoil Factor 2 Sensitizes Rat Pups to Systemic Infection with the Neonatal Pathogen Escherichia coli K1
Collaboration with Peter Taylor’s lab at UCL published in Infection & Immunity identifies Tff2 as a key factor preventing systemic dissemination of neuropathogenic E. coli in neonates
The Nlrp6 inflammasome is not required for baseline colonic inner mucus layer formation or function
Joana Volk’s JEM paper delineates the relative roles of the inflammasome and the microbiota in shaping the barrier function of the colonic mucus layer
2018
Calcium-activated Chloride Channel Regulator 1 (CLCA1) Controls Mucus Expansion in Colon by Proteolytic Activity
Collaboration with Malin Johansson’s lab published in EBioMedicine reveals the role of goblet cell secreted CLCA1 as an important regulator of mucus processing
Bifidobacteria or Fiber Protects against Diet-Induced Microbiota-Mediated Colonic Mucus Deterioration
Collaboration with Fredrik Bäckhed’s and Gunnar Hansson’s labs published in Cell Host & Microbe characterizes how exposure to a low fibre, high fat diet results in a compromised colonic mucus barrier function
2016-17
Postnatal development of the small intestinal mucosa drives age-dependent, regio-selective susceptibility to Escherichia coli K1 infection
Study published in Scientific Reports showing how neuropathogenic E. coli exploit underdeveloped neonatal goblet cell-intrinsic defences to invade the small intestine
A sentinel goblet cell guards the colonic crypt by triggering Nlrp6-dependent Muc2 secretion
Study published in Science describing and characterizing a novel sub-population of colonic goblet cells that integrate innate immune signalling pathways to regulate mucus secretion in response to bacteria
Gram-positive bacteria are held at a distance in the colon mucus by the lectin-like protein ZG16
Investigation published in PNAS characterizing the goblet cell-secreted ZG16 as a peptidoglycan-binding protein that aggregates Gram-positive bacteria and prevents them penetrating into the colonic mucus barrier
Open positions
The lab is always willing to consider postdoctoral applicants who are seeking to apply for their own postdoctoral grants from external funders (e.g. VR, SSMF, Cancerfonden, Marie Curie, HFSP etc). If you have an interest in our work and would like to propose your own postdoctoral project, you are welcome to get in touch and we will assist you as much as possible in the application.
Essentials:
Doctoral degree (or equivalent) in a relevant subject area (e.g. Biology, Biotechnology, Biochemistry, Pharmacy, Medicine etc.)
Demonstrable skills in written and spoken English
Register your interest:
Potential candidates should contact George for more details. Please include a cover letter (max 1 page of A4), CV and up to 2 references.
Postdoctoral researchers
(2-4 years full time employment)
The lab often has potential opportunities for motivated students who would like to gain some research experience during the summer break, or as part of Bachelors or Masters level degree research projects. Please contact George for more details and include a cover letter (max 1 page of A4) and a CV describing all previous laboratory and methodological experience.
Undergraduate student projects
The lab is grateful to be supported by past and ongoing infrastructure and funding from:

The cluster of new and established groups originally founded by Prof. Gunnar C. Hansson at University of Gothenburg



Wallenberg Centre for Molecular & Translational Medicine


Contact:
Email: george.birchenough@gu.se
Office phone: +46(0)723319626
Visiting address: The Lundberg Laboratory, Medicinaregatan 9C, 413 90 Gothenburg, Sweden
Postal address: Box 440, 405 39 Gothenburg, Sweden